Decentralized Clinical Trials Market Analysis Driving Digital Transformation in Research
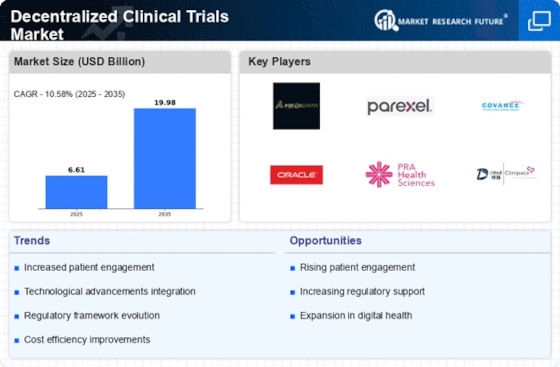
The Decentralized Clinical Trials Market Analysis highlights the rapid transformation occurring in clinical research due to digital technologies and remote patient monitoring. Traditional clinical trials often require participants to travel to research sites for testing and monitoring, which can limit participation and slow recruitment. Decentralized clinical trials address these challenges by integrating telemedicine, wearable devices, mobile health applications, and electronic patient reporting tools. These technologies enable researchers to collect real-time patient data without requiring participants to visit a centralized trial site frequently. As pharmaceutical companies and research organizations increasingly adopt digital solutions, decentralized trials are becoming a preferred approach for improving efficiency, reducing costs, and accelerating drug development timelines. Furthermore, patient-centric trial models are gaining popularity because they allow individuals to participate from their homes, significantly increasing accessibility and diversity in clinical studies.
The market is also expanding because regulatory authorities worldwide are encouraging the adoption of innovative clinical trial models. Healthcare organizations are investing heavily in digital health infrastructure to support remote data collection, data security, and patient engagement platforms. Another factor influencing the decentralized clinical trials market is the increasing use of artificial intelligence and data analytics to manage large datasets collected from remote devices. AI-powered platforms help researchers analyze patient responses faster and identify trends that can guide clinical decision-making. Pharmaceutical companies are also collaborating with technology providers to design flexible clinical trial frameworks that combine both decentralized and traditional approaches. This hybrid model allows sponsors to maintain data quality while benefiting from improved patient engagement and faster recruitment. As the demand for efficient research methodologies grows, decentralized trials are expected to remain a critical component of modern clinical development strategies.
FAQ
Q1. What are decentralized clinical trials?
Decentralized clinical trials use digital technologies and remote monitoring to conduct clinical research without requiring patients to visit a central site frequently.
Q2. Why are decentralized trials becoming popular?
They improve patient convenience, increase recruitment diversity, reduce operational costs, and accelerate drug development.
Q3. Which technologies support decentralized trials?
Technologies include telemedicine platforms, wearable health devices, mobile health apps, and remote patient monitoring systems.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Παιχνίδια
- Gardening
- Health
- Κεντρική Σελίδα
- Literature
- Music
- Networking
- άλλο
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness
